Is sodium a very reactive metal
Is Sodium A Very Reactive Metal. â prev question next question â 1 vote. The free metal does not occur in nature and must be prepared from compounds. Sodium is a chemical element with the symbol na from latin natrium and atomic number 11. Its only stable isotope is 23 na.

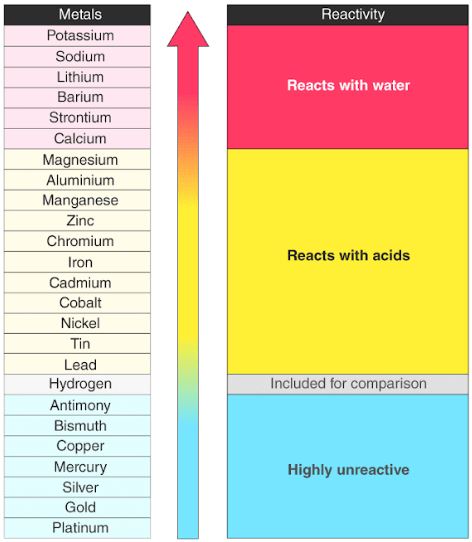
Avoid any contact with skin as the hot lye causes severe burns. Sodium is a chemical element with the symbol na from latin natrium and atomic number 11. This metal is very reactive has properties similar to those of magnesium and is in the same period as bromine. Sodium is one of the most reactive metals that. Sodium losses an electron and gains a positive charge. As a part of the alkali metal group in the periodic table sodium is highly reactive and is never found pure in nature.
Sodium is so reactive due to two main factors.
Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. Its only stable isotope is 23 na. â prev question next question â 1 vote. C copper displaces zinc from zinc sulphate solution. Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. Add your answer and earn points.
 Source: quora.com
Source: quora.com
Its only stable isotope is 23 na. Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. As a part of the alkali metal group in the periodic table sodium is highly reactive and is never found pure in nature. It tarnishes easily and has a low melting point and density. There is a reaction would take place between magnesium and sodium chloride.
 Source: chegg.com
Source: chegg.com
Sodium is so reactive because they lose their valence electron very easily. Add your answer and earn points. Sodium losses an electron and gains a positive charge. It tarnishes easily and has a low melting point and density. Avoid any contact with skin as the hot lye causes severe burns.

C copper displaces zinc from zinc sulphate solution. Sodium is one of the most reactive metals that. Avoid any contact with skin as the hot lye causes severe burns. â prev question next question â 1 vote. The element chlorine figure pageindex 1b is a pale yellow corrosive gas that should not be inhaled due to its poisonous nature.
 Source: pinterest.co.uk
Source: pinterest.co.uk
The answer to this question is yes. There is a reaction would take place between magnesium and sodium chloride. Sodium like all the group 1 metals is very reactive because it wants to achieve what is called an inert gas electron configuration. Its only stable isotope is 23 na. Sodium is so reactive because they lose their valence electron very easily.
 Source: richardbowles.co.uk
Source: richardbowles.co.uk
There is a reaction would take place between magnesium and sodium chloride. Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. Sodium is a very reactive metal. This metal is very reactive has properties similar to those of magnesium and is in the same period as bromine. As a part of the alkali metal group in the periodic table sodium is highly reactive and is never found pure in nature.
 Source: byjus.com
Source: byjus.com
Sodium is so reactive due to two main factors. Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. Sodium is a chemical element with the symbol na from latin natrium and atomic number 11. Sodium is one of the most reactive metals that. C copper displaces zinc from zinc sulphate solution.

The chemical reaction between these two metals is given below. Sodium like all the group 1 metals is very reactive because it wants to achieve what is called an inert gas electron configuration. This metal is very reactive has properties similar to those of magnesium and is in the same period as bromine. Sodium is so reactive due to two main factors. Sodium is so reactive because they lose their valence electron very easily.
 Source: thoughtco.com
Source: thoughtco.com
Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. It is a soft silvery white highly reactive metal sodium is an alkali metal being in group 1 of the periodic table. â prev question next question â 1 vote. It is because sodium is one of the most reactive metals. Sodium is so reactive because they lose their valence electron very easily.
 Source: dynamicscience.com.au
Source: dynamicscience.com.au
As a part of the alkali metal group in the periodic table sodium is highly reactive and is never found pure in nature. â prev question next question â 1 vote. Sodium like all the group 1 metals is very reactive because it wants to achieve what is called an inert gas electron configuration. Sodium is one of the most reactive metals that. Avoid any contact with skin as the hot lye causes severe burns.
 Source: brainly.in
Source: brainly.in
The answer to this question is yes. The free metal does not occur in nature and must be prepared from compounds. Both can form hydroxides when reacted with water. â prev question next question â 1 vote. Sodium is so reactive because they lose their valence electron very easily.
 Source: toppr.com
Source: toppr.com
There is a reaction would take place between magnesium and sodium chloride. Sodium losses an electron and gains a positive charge. Sodium is so reactive because they lose their valence electron very easily. â prev question next question â 1 vote. The element chlorine figure pageindex 1b is a pale yellow corrosive gas that should not be inhaled due to its poisonous nature.

Its only stable isotope is 23 na. Avoid any contact with skin as the hot lye causes severe burns. Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water. This metal is very reactive has properties similar to those of magnesium and is in the same period as bromine. Sodium is a very reactive metal.
 Source: youtube.com
Source: youtube.com
Add your answer and earn points. Sodium like all the group 1 metals is very reactive because it wants to achieve what is called an inert gas electron configuration. Its only stable isotope is 23 na. It is in fact second in the reactivity table of elements. This metal is very reactive has properties similar to those of magnesium and is in the same period as bromine.
 Source: slideshare.net
Source: slideshare.net
The element chlorine figure pageindex 1b is a pale yellow corrosive gas that should not be inhaled due to its poisonous nature. It is a soft silvery white highly reactive metal sodium is an alkali metal being in group 1 of the periodic table. There is a reaction would take place between magnesium and sodium chloride. Sodium losses an electron and gains a positive charge. It is because sodium is one of the most reactive metals.
 Source: chegg.com
Source: chegg.com
There is a reaction would take place between magnesium and sodium chloride. The free metal does not occur in nature and must be prepared from compounds. As a part of the alkali metal group in the periodic table sodium is highly reactive and is never found pure in nature. It is because sodium is one of the most reactive metals. Yes sodium is very reactive metal just because of this it is kept in kerosene or some another oil because it is very reactive with oxygen and water.
If you find this site serviceableness, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title is sodium a very reactive metal by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.